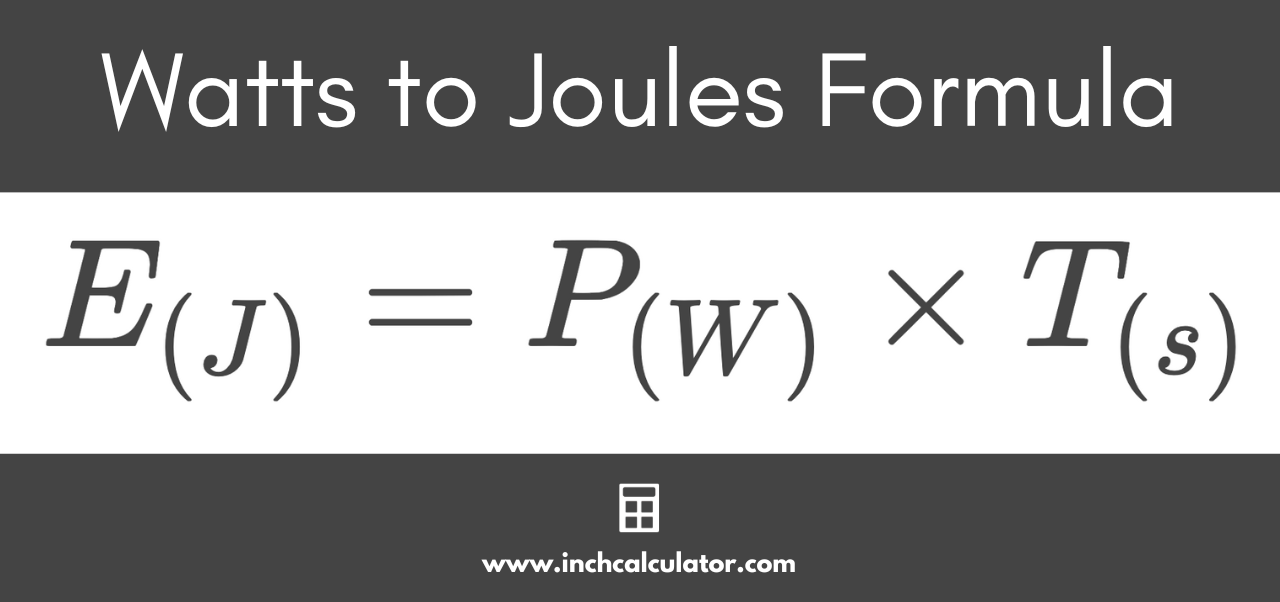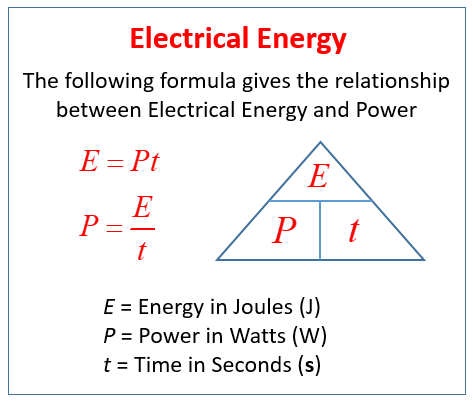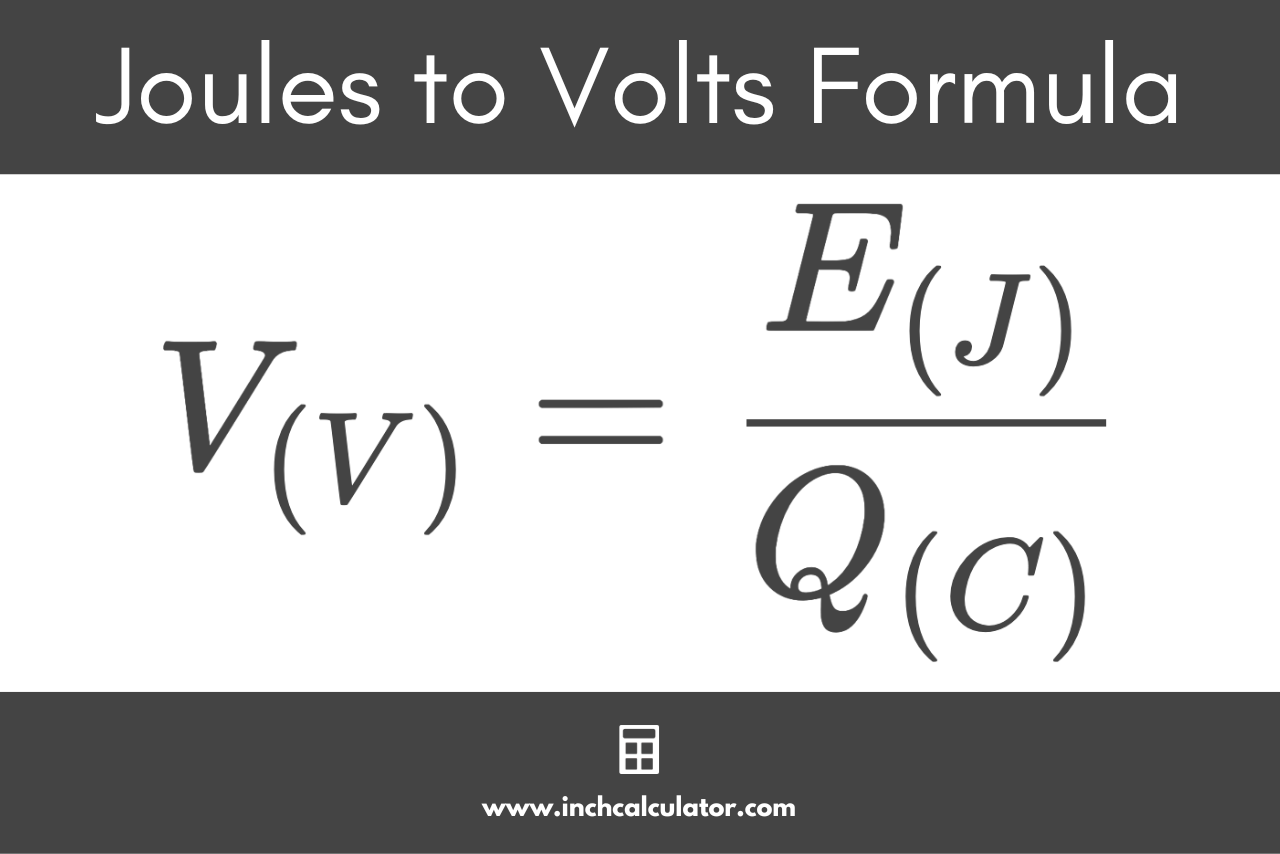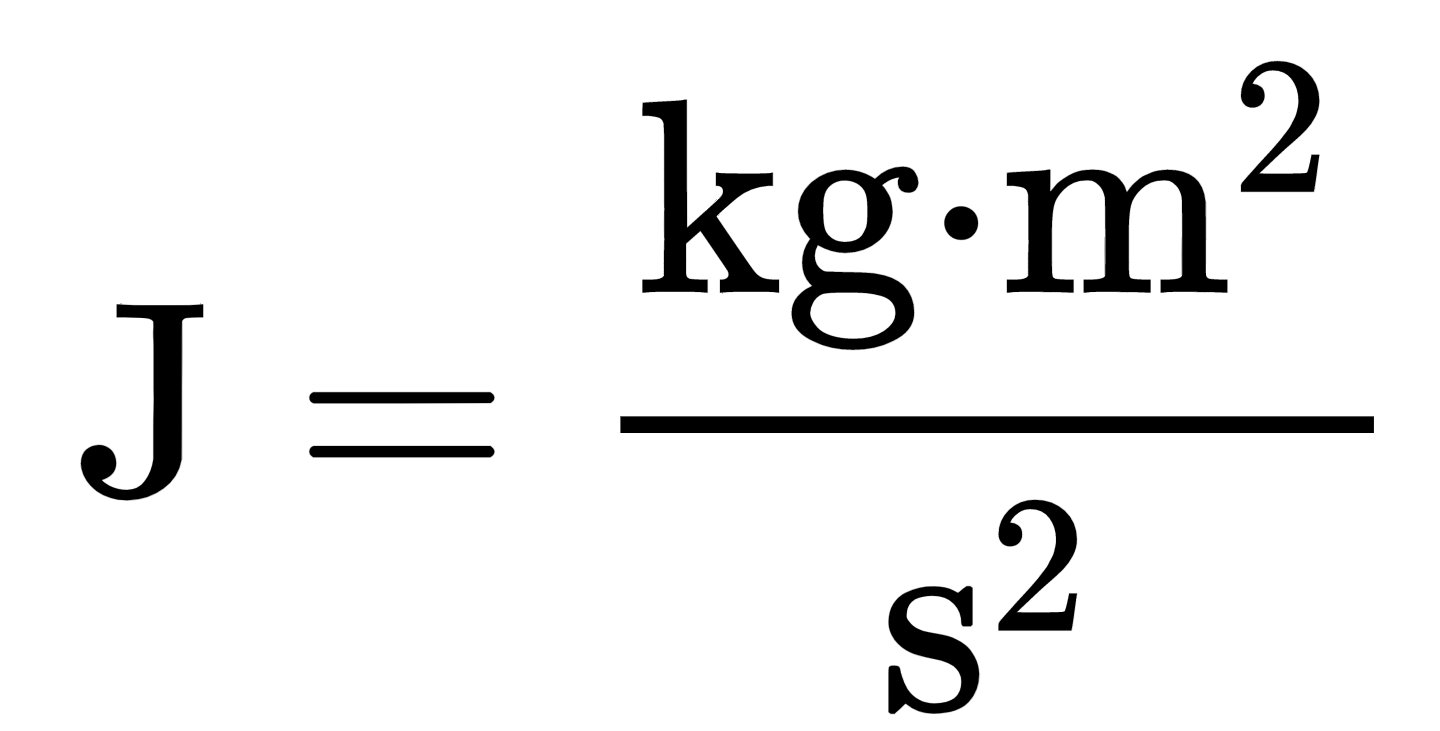
cyber wolf35 on X: "@ArgonCapital Anyway to finish what I was saying above when you use the provided equations to solve for the kilogram your answer comes out in joules, look at

SOLVED: Calculate the heat capacity, in joules and in calories per degree, of the following: (a) 28.4 g of water (b) 1.00 oz of lead