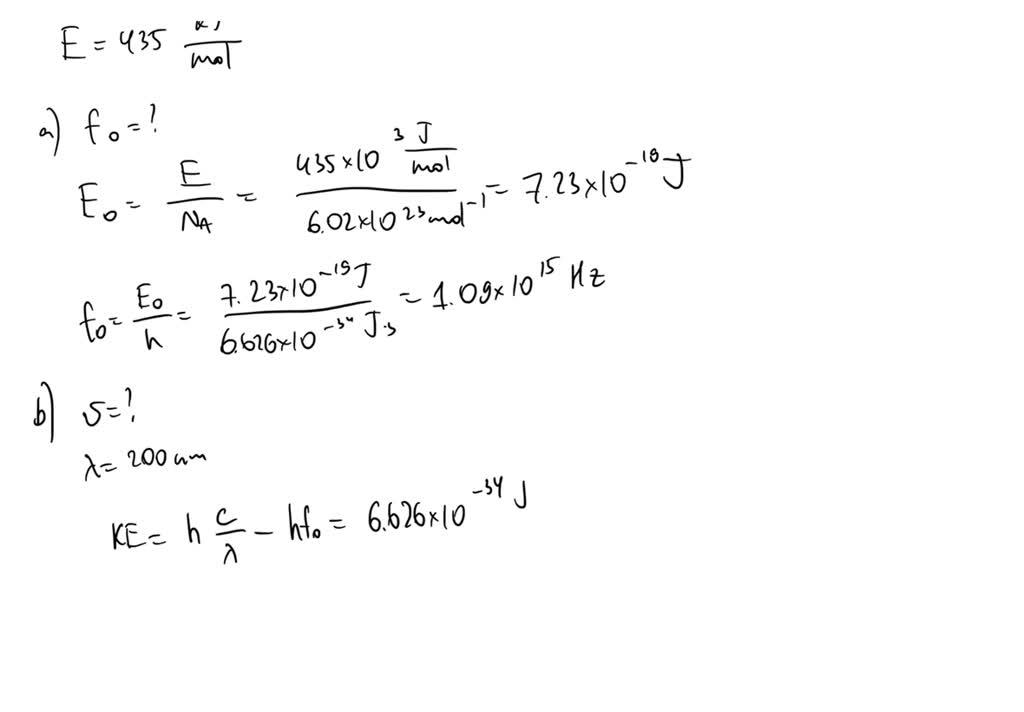
SOLVED: The binding energy for mercury is 435 kJ/mol of photons. (a) Calculate the threshold frequency. (b) Calculate the maximum velocity of the electrons ejected when light of wavelength 200 nm strikes
the threshold frequency for a metal is 6.2×10.^3 8-1.calculate KE emitted of an electron when the radiation - Brainly.in

Electrons are emitted with zero velocity from a metal surface when it is exposed to radiation of wavelength 6800 ∘A . Calculate threshold frequency v0 and work function w0 of the metal.

Photoelectric threshold frequency of a metal is V0 .If light frequency of 4 V0 . is incident on it, the maximum K.E. of the ejected photoelectron is.

Electrons are emitted with zero velocity from metal surface when it is exposed to radiation of wave¬length 6800 A. Calculate threshold frequency (v0) and work function (W0) of the metal - CBSE
Threshold frequency of a certain metal for photoelectric emission is 5 × 10^14 Hz. Calculate its work function. - Sarthaks eConnect | Largest Online Education Community

CHEM 101: Photoelectric Effect - Threshold Frequency and Binding Energy from Kinetic Energy - YouTube
calculate the threshold frequency of the metal from which the photoelectrons are emitted with zero velocity when exposed to radiation of wavelength 6800 Armstrong
If light of threshold frequency falls on a metal surface then Kinetic Energy of ejected electron must be zero, is it true? - Quora

Finding the Frequency Threshold of Incident Light for Electron Emission from a Material with Given Work Function | Physics | Study.com
Threshold frequency of copper is 1.12 × 10^15 Hz. Light of wavelength 253.7Å is incident on its surface. - Sarthaks eConnect | Largest Online Education Community