SOLVED: A better and slightly more tedious calculation uses the 0512 Vu Extended Debye-Huckel equation log Y = √(1 + 305) with the ion size √(900 pm) listed for H in Table

Accuracy of the Debye-Hückel limiting law Example: The mean activity coefficient in a mol kg -1 MnCl 2 (aq) solution is 0.47 at 25 o C. What is the. - ppt download

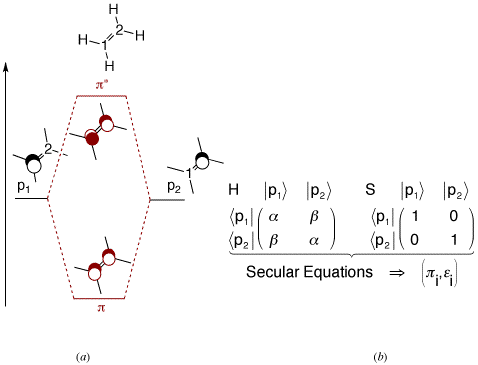
computational chemistry - Tight binding model calculation with Extended Huckel Approximation - Computational Science Stack Exchange